A mechanistic understanding of the degradation of trace organic contaminants by UV/hydrogen peroxide, UV/persulfate and UV/free chlorine for water reuse†
Abstract
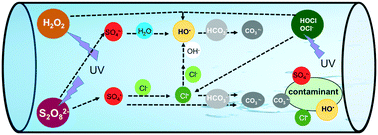
Climate change and population growth pose increasing challenges to the availability of freshwater resources. Ultraviolet-based advanced oxidation processes (UV/AOPs) are promising water reuse technologies that increase drinking water supplies through wastewater reuse. This study examined the fundamental mechanisms of contaminant degradation and reactive species generation in three UV/AOPs, i.e., UV/hydrogen peroxide (H2O2), UV/persulfate (S2O82−) and UV/chlorine (HOCl), on the basis of combined experimental investigation and kinetic modeling on the degradation of six representative trace organic contaminants. The formation and distribution of reactive radicals including hydroxyl radical (HO˙), sulfate radical (SO4˙−), and chlorine atom (Cl˙) were investigated. Results showed that the treatment efficiency of contaminants generally followed the order UV/S2O82− > UV/H2O2 > UV/HOCl under chemical conditions relevant to treatment train steps utilized in water reuse. The generation of HO˙ was important in UV/H2O2, whereas both SO4˙− and HO˙ were important in UV/S2O82− and CO3˙− predominated in UV/HOCl. Among the three UV/AOPs, the treatment efficiency of UV/S2O82− was most sensitive to pH, chloride, and inorganic carbon. The combined experimental and modeling approach provided guidance for the design and optimization of UV/AOP systems for water reuse under diverse chemical conditions.
- This article is part of the themed collections: Environmental Science: Water Research & Technology: Editors' Choice and Environmental Science: Water Research & Technology 2017 Most Downloaded Articles

 Please wait while we load your content...
Please wait while we load your content...