X-ray absorption fine structure study of enhanced sequestration of U(vi) and Se(iv) by montmorillonite decorated with zero-valent iron nanoparticles†
Abstract
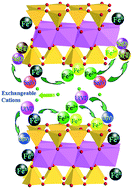
Herein, using Na-montmorillonite and Al-montmorillonite as templates, supported NZVI with superior reactivity, namely NZVI/Na-Mont and NZVI/Al-Mont, was synthesized and applied for enhanced sequestration of U(VI) and Se(IV). The results indicated that the negatively charged Na-Mont can effectively adsorb cationic U(VI), while the positively charged Al-Mont can adsorb anionic Se(IV), significantly enhancing the rate and extent of U(VI) and Se(IV) sequestration on NZVI due to the synergistic effect between adsorption and reduction. The inhibition effect of 1,10-phenanthroline demonstrated that surface-adsorbed Fe(II) on corrosion products and clay surfaces played an indispensable role in U(VI) and Se(IV) reduction, which was further confirmed by XPS identification. XANES analysis demonstrated that in the NZVI system, Na-Mont and Al-Mont promote the reductive transformation of U(VI) into U(IV), and Se(IV) into Se(0)/Se(−II), respectively. EXAFS analysis indicated the presence of Al/Si scattering for U(VI)-treated NZVI/Na-Mont and Se(IV)-treated NZVI/Al-Mont samples, revealing that Na-Mont and Al-Mont reacted as a scavenger for insoluble products like UO2 and FeSe, thereby, more reactive sites can be used for U(VI) and Se(IV) reduction. Our results suggested that the distinct structure of modified montmorillonite can be utilized for synthesizing supported NZVI to create properties compatible for enhanced enrichment of radionuclides, displaying potential application in nuclear waste management.

 Please wait while we load your content...
Please wait while we load your content...