Galvanic-like cells produced by negative charge nonuniformity of lattice oxygen on d-TiCuAl–SiO2 nanospheres for enhancement of Fenton-catalytic efficiency†
Abstract
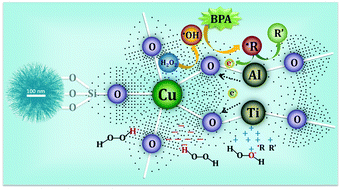
An efficient Fenton-type nano-catalyst consisting of Cu, Ti and Al lattice-doped dandelion-like silica nanospheres (d-TiCuAl–SiO2 Ns) was successfully prepared via a hydrothermal process for the first time. The lattice substitution of Cu, Ti and Al for Si initiated a higher and a lower electron density of the lattice O2− around the lattice Cu and near the lattice Ti & Al, respectively, through the higher electronegativity of Cu on the basis of extended X-ray absorption fine structure, X-ray photoelectron spectroscopy, Fourier-transform infrared spectroscopy, cyclic voltammetry and electron paramagnetic resonance measurement. Thus, countless surface galvanic-like cells formed between the electron-rich Cu center (cathode) and the electron-deficient Ti and Al region (anode) on the nanospheres, enhancing the selective adsorption of H2O2 on d-TiCuAl–SiO2 Ns. This kind of surface structure facilitated the reduction of H2O2 to ˙OH at the cathode and induced the oxidation of organic radical intermediates (˙R) on the anode through the delivery of the electron of ˙R to the cathode during the Fenton reaction, which resulted in almost all of the energy of H2O2 being applied to the degradation of pollutants. Therefore, d-TiCuAl–SiO2 Ns were highly effective and stable for persistent contaminant degradation at neutral pH values, which was ∼10 times higher than that of the conventional Fenton catalysts. Moreover, the utilization efficiency of H2O2 has been maintained at a very high level throughout the Fenton reaction. Our findings indicated that the special surface design of nanoparticles could be applied toward the improvement of Fenton reaction efficiency and utilization of H2O2.

 Please wait while we load your content...
Please wait while we load your content...