Reactivity of ferrihydrite and ferritin in relation to surface structure, size, and nanoparticle formation studied for phosphate and arsenate†
Abstract
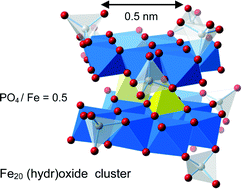
Ferritin (Ftn) is a natural protein that can store metal (hydr)oxide nanoparticles of tunable size in its cavity and bind oxyanions. This quality can be used in water purification by applying nanotechnology. As our study suggests, the adsorption behavior of engineered ferritin strongly resembles that of ferrihydrite (Fh) which is in nature the most insoluble and stable Fe (hydr)oxide at the nanoscale due to its exceptionally low surface Gibbs free energy. For freshly prepared Fh, the adsorption of phosphate has been measured and modeled as a function of pH (4–11), solution concentration (μM–mM), and background electrolyte level (0.01–1 M). In connection to surface complexation modeling (SCM) the surface structure of Fh has been analyzed, quantifying the unique polyhedral surface composition with singly coordinated surface groups that may form either monodentate or bidentate surface complexes. The spherical double layer theory has been implemented for downscaling to very small cluster sizes, which is vital for studying nanoparticle behavior in natural as well as synthetic systems. Modeling suggests the formation of very small Fh clusters when particles are produced in the absence of Ftn by instantaneous Fe(III) neutralization, leading at pH 7 on average to 62 ± 3 Fe per particle with a solubility product of log Qso = −37.3 ± 0.2. The corresponding size of the critical nucleus (nFe = 11 ± 2) matches well with the smallest imaginable Fh cluster with a Fe13 Keggin structure. Application of the model to engineered ferritin (Ftn) suggests that in the initial step of preparation, more than one Fe (hydr)oxide cluster per ferritin can be formed, having a very small size (nFe ∼17). The initial clusters, metastable in Ftn, can grow at stepwise additions of Fe and will fuse to a large singlet above ∼1000 Fe per Ftn. However, the clusters remain very small (∼20 ± 5 Fe) when Ftn is loaded with Fe in the presence of PO4. These ultra-small clusters are stabilized by the adsorption of PO4 (∼5–10 PO4 per cluster) that decreases the surface Gibbs free energy (Gsurf). At a large decrease in Gsurf due to ion adsorption, Fe-oxyanion networks will form that can be important in terrestrial and aquatic systems.

 Please wait while we load your content...
Please wait while we load your content...