Interactions between 2,4-bis-pteridine-1,5-benzodiazepine and group 12 dihalides: synthesis, spectral and XRD structural studies and theoretical calculations†
Abstract
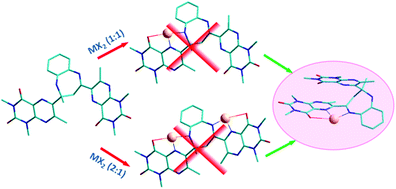
2,4-Bis(1,3,7-trimethyl-pteridine-2,4(1H,3H)-dione-6-yl)-2,3-dihydro-2-methyl-1H-1,5-benzodiazepine (DLMBZD) has been prepared and its molecular and crystal structures have been determined from spectral and XRD data. The benzodiazepine ligand was reacted with zinc(II), cadmium(II) and mercury(II) chloride, bromide and iodide to give complexes with general formula [M(DLMBZD)X2]. The complexes have been synthesized and characterized by IR, NMR and elemental analysis. The structure of seven complexes has been obtained by single crystal X-ray diffraction. In all the cases, the metal is (2 + 2 + 1)-five-coordinated by two halide ligands, two nitrogen atoms from pyrazine and diazepine rings and a carbonyl oxygen from a pteridine ring. The coordinated-metal environment is a square-based pyramid, with increasing trigonality from Hg(II) to Zn(II) complexes. To coordinate the metals, the ligand folds itself, establishing four intramolecular σ–π interactions with the pyrimidine and pyrazine rings. A topological analysis of the electron density using the Quantum Theory of Atoms in Molecules and the complexes stability has been performed.


 Please wait while we load your content...
Please wait while we load your content...