Revisiting oxo-centered carbonyl-triruthenium clusters: investigating CO photorelease and some spectroscopic and electrochemical correlations†
Abstract
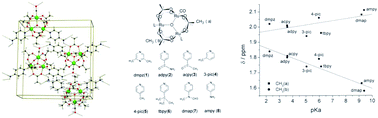
We synthesized and characterized a series of oxo-centered carbonyl-triruthenium complexes with the general formula [Ru3O(CH3COO)6(L)2(CO)], where L = 2,6-dimethylpyrazine (dmpz) (1), isonicotinamide (adpy) (2), 4-acetylpyridine (acpy) (3), 3-methylpyridine (3-pic) (4), 4-methylpyridine (4-pic) (5), 4-tert-butylpyridine (4-tbpy) (6), 4-(dimethyl)aminopyridine (dmap) (7), or 4-aminopyridine (ampy) (8); we also investigated the photoreactivity of these complexes. Single-crystal X-ray diffraction helped to elucidate the structures of 1·H2O, 7·C2H4Cl2, and 8. The unit cell of 8 is composed of four cluster units; the hydrogen bonds between the amino groups of the terminal ligand of a neighboring molecule and the oxygen atoms of CO or acetate bridging ligands hold these cluster units together. The spectroscopic (NMR, UV-visible, and IR) and the electrochemical properties (cyclic voltammetry) of these complexes correlated with the ancillary ligands in terms of their σ-donating and π-accepting characteristics. The molecular orbital and the electronic localized description of the [Ru3O]–CO unit helped to rationalize the correlations. The photoreactivity of compounds 1–8 was investigated by laser excitation at 377 nm. Given the CO photorelease quantum yields, σ-donor ligands and aqueous medium (more polar) stabilized the charge-transfer excited state that culminated in CO photosubstitution, leading to higher Φ values.


 Please wait while we load your content...
Please wait while we load your content...