Photoswitchable Cu II4MoIV and Cu II2MoIV cyanido-bridged molecules†
Abstract
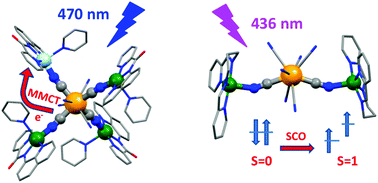
The self-assembly of copper(II) complexes with two Schiff base ligands: L1O = N3 and L2 = N4 and octacyanidomolybdate(IV) ions yields two discrete molecules of odd nuclearity, namely pentametallic [Cu(L1O)(py)]4[MoIV(CN)8]·14H2O (1), Cu4Mo and trimetallic [Cu(L2)]2[MoIV(CN)8]·9H2O (2), Cu2Mo. Both molecular systems have been characterised structurally and magnetically, revealing a photomagnetic effect. In the case of (1) a metal-to-metal charge transfer (MMCT) mechanism is proposed. The analysis of magnetic interactions in the photogenerated state of (1) assumes the formation of the metastable cluster CuII3CuIMoV where metal centres in MoV–CN–CuII linkages are ferromagnetically coupled with J2 = 104(3) cm−1. In (2) the increase in the magnetisation is due to the singlet–triplet transition on the MoIV centre leading to the formation of the metastable CuII2MoIVHS. The presence of the paramagnetic MoIVHS (S = 1) centre linking the CuII (S = 1/2) centres allows for effective ferromagnetic interaction of 3 paramagnetic centres with coupling constant J2 = 20.2(7) cm−1.
- This article is part of the themed collection: Molecular Spintronics : The role of Coordination Chemistry

 Please wait while we load your content...
Please wait while we load your content...