Cyclopalladated organosilane–tethered thiosemicarbazones: novel strategies for improving antiplasmodial activity
Abstract
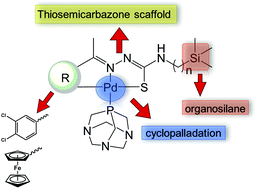
Two series of ferrocenyl- and aryl-derived cyclopalladated organosilane thiosemicarbazone complexes were synthesised via C–H bond activation. Selected compounds were evaluated for in vitro antiplasmodial activity against the chloroquine-sensitive (NF54) and chloroquine-resistant (Dd2) strains of the human malaria parasite Plasmodium falciparum. Cyclopalladation of the thiosemicarbazones resulted in antiplasmodial activities in the low micromolar range.

 Please wait while we load your content...
Please wait while we load your content...