Blending materials composed of boron, nitrogen and carbon to transform approaches to liquid hydrogen stores†
Abstract
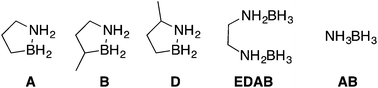
Mixtures of hydrogen storage materials containing the elements of boron, nitrogen, carbon, i.e., isomers of BN cyclopentanes are examined to find a ‘fuel blend’ that remains a liquid phase throughout hydrogen release, maximizes hydrogen storage density, minimizes impurities and remains thermally stable at ambient temperatures. We find that the mixture of ammonia borane dissolved in 3-methyl-1,2-dihydro-1,2-azaborolidine (compound B) provide a balance of these properties and provides ca. 5.6 wt% hydrogen. The two hydrogen storage materials decompose at a faster rate than either individually and products formed are a mixture of molecular trimers. Digestion of the product mixture formed from the decomposition of the AB + B fuel blend with methanol leads to the two corresponding methanol adducts of the starting material and not a complex mixture of adducts. The work shows the utility of using blends of materials to reduce volatile impurities and preserve liquid phase.
- This article is part of the themed collection: Main Group Transformations

 Please wait while we load your content...
Please wait while we load your content...