Genesis of a bi-functional acid–base site on a Cr-supported layered double hydroxide catalyst surface for one-pot synthesis of furfurals from xylose with a solid acid catalyst†
Abstract
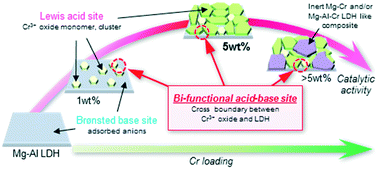
A Cr-supported Mg–Al layered double hydroxide (Cr/Mg–Al LDH) comprised bi-functional Lewis acid–Brønsted base active sites on the catalyst surface at close boundaries between Cr3+ oxide and Mg–Al LDH. These enabled efficient aldose–ketose isomerization. The combined use of solid Brønsted acid Amberlyst-15 and bi-functional acid–base Cr/Mg–Al LDH exhibits higher activity for one-pot transformation of xylose into furfural via aldose–ketose isomerization and successive dehydration under mild conditions than those exhibited by basic bare Mg–Al LDH, substituted Mg–Cr LDH, and Lewis acidic Cr3+ supported catalysts with Amberlyst-15. The correlation between the activity and the Cr loading amount for Mg–Al LDH was evaluated using several characterization techniques and MPV reduction as a model reaction for Lewis acidity investigation. The results show the following: (1) the monomer of Lewis acidic Cr3+ oxide is supported on the Mg–Al LDH surface below 1 wt%. (2) Thereafter, Cr3+ species form small Lewis acidic Cr3+ oxide dimers and/or trimers up to 5 wt%. Then, they cover the Mg–Al LDH surface to generate highly active bi-functional Lewis acid–Brønsted base sites. (3) Above 5 wt%, the excess Cr3+ species generate inert Mg–Cr and/or Mg–Al–Cr LDH-like composites. They are deposited on some active sites, leading to decreased activity. The results show that furfural yields vary significantly in accordance with Cr loadings and that the 5 wt% Cr/Mg–Al LDH achieved the highest value (59% yield, 18 h) because the most effective interaction between the Lewis acidic Cr3+ oxide and the Mg–Al LDH basic site is observed at 5 wt% Cr loading.

 Please wait while we load your content...
Please wait while we load your content...