A general protocol for the reductive N-methylation of amines using dimethyl carbonate and molecular hydrogen: mechanistic insights and kinetic studies†
Abstract
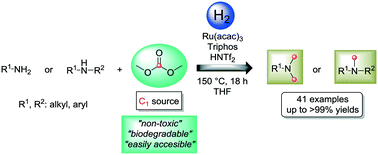
Herein, we report for the first time a general and selective ruthenium-catalyzed reductive methylation of amines using dimethyl carbonate as a C1 source and molecular hydrogen as a reducing agent. Notably, this methodology allows N-methylated tertiary aromatic and aliphatic amines to be obtained with good to excellent yields using a green, non-toxic and biodegradable carbon source in the presence of an in situ formed Ru/Triphos complex. The catalytic protocol presented here opens the possibility of developing new sustainable processes for the selective synthesis of N-methyl substituted amines using molecular hydrogen. Mechanistic and kinetic studies have been carried out in order to understand the pathways involved in the general reaction mechanism for the N-methylation of aniline.

 Please wait while we load your content...
Please wait while we load your content...