Selective dehydrogenation of bioethanol to acetaldehyde over basic USY zeolites†
Abstract
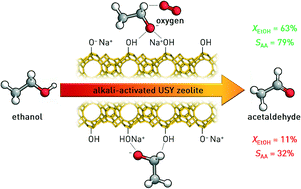
Zeolites featuring basic sites have displayed outstanding performance in the valorisation of biobased substrates through condensation and coupling reactions. Herein, we present the continuous-flow, gas-phase dehydrogenation of ethanol to acetaldehyde as a novel application of the recently developed alkali-activated high-silica USY zeolites. Evaluation of the hydroxides of Group 1 metals in the catalyst preparation by alkaline treatment identified sodium hydroxide as the most suitable base. This is due to the stronger character and the higher number of mildly basic sites formed coupled with the minimal impact on the pristine porous properties of the zeolite, which resulted in superior catalytic performance. Compared to existing basic catalysts such as MgO and hydroxyapatites, no C4-condensation products were observed and the occurrence of ethanol dehydration to ethylene was marginal due to the negligible acidity of the material. At the optimal reaction temperature, acetaldehyde was attained with a ca. 15-fold higher yield (50%) and with high selectivity (80%) when adding oxygen to the ethanol feed. The origin of this peculiar positive behaviour was unraveled through kinetic and in situ infrared spectroscopic studies as well as by the temperature-programmed surface reaction of ethanol. These investigations revealed the exclusive presence of ethoxide species at the catalyst surface, which (i) are formed upon adsorption of ethanol at basic siloxy groups, (ii) are stabilised through hydrogen bonding with vicinal silanols and (iii) react in a rate-limiting step with gas-phase oxygen through an Eley–Rideal mechanism to form acetaldehyde, thus releasing water. In contrast to the rapid deactivation shown by noble metal based catalysts, the material exhibited stable performance over 24 h on stream.
- This article is part of the themed collection: Catalysis on Zeolites

 Please wait while we load your content...
Please wait while we load your content...