Improvement in the catalytic properties of ZSM-5 zeolite nanoparticles via mechanochemical and chemical modifications†
Abstract
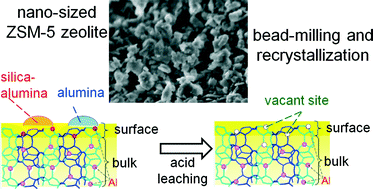
Improving the selectivity of zeolite catalysts over a wide variety of reactions without diminution of their catalytic activity requires both shortening the diffusion path of guest molecules within the micropores and selective deactivation of the external acid sites. In this work, the former was achieved by mechanochemical bead-milling treatment of micron-sized ZSM-5 zeolite and subsequent recrystallization, and the latter by the selective removal of framework Al and silica–alumina species on the external surface of ZSM-5 nanoparticles via hydrothermal treatment with a concentrated HNO3 aqueous solution. The modified ZSM-5 nanoparticles showed higher propylene yields as well as high resistance to coke formation during hexane cracking. Significant changes in the Brønsted and Lewis acid sites on the external surface of the treated ZSM-5 were also observed using FT-IR with CO and collidine as probe molecules.
- This article is part of the themed collection: Catalysis on Zeolites

 Please wait while we load your content...
Please wait while we load your content...