Preparation of high oxygen storage capacity and thermally stable ceria–zirconia solid solution
Abstract
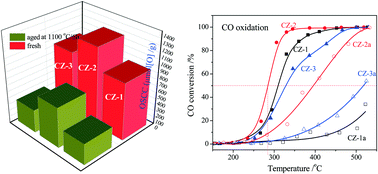
Ceria–zirconia solid solution is a very important material in three-way catalysts for automotive emission control. High oxygen storage capacity (OSC) and thermally stable Ce0.5Zr0.5O2 was prepared by a modified complexing–coprecipitation (CC) method, and its surface area reached 44 m2 g−1 after calcination at 1100 °C for 6 h. Based on the characterizations of its structural and physicochemical properties, it was found that Ce0.5Zr0.5O2 prepared by the CC method existed as the t′′-phase with rich oxygen defects and surface Ce3+ and has a larger BET surface area, uniform particle and pore sizes, and excellent bulk oxygen migration and redox abilities than samples prepared by other methods. After being calcined at 1100 °C for 6 h, its surface area, OSC (and OSCC, oxygen storage capacity complete) and catalytic activity for the oxidation of CO were still the best among three Ce0.5Zr0.5O2 solid solutions prepared by three methods whether it was used as the catalyst or as a support for a Pd catalyst, reflecting its good thermostability, although its particle and pore sizes were somewhat increased. This complexing–coprecipitation method can be used to prepare other high surface area and thermally stable inorganic materials.
- This article is part of the themed collection: 2016 most accessed Catalysis Science and Technology articles

 Please wait while we load your content...
Please wait while we load your content...