Lewis acidic zeolite Beta catalyst for the Meerwein–Ponndorf–Verley reduction of furfural†
Abstract
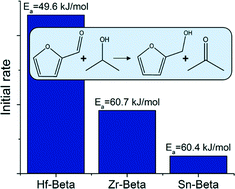
The catalytic properties of tin-, zirconium- and hafnium-containing siliceous zeolite Beta were investigated in the Meerwein–Ponndorf–Verley reduction of furfural using a liquid-phase plug-flow microreactor at temperatures between 328–358 K. Catalytic rates varied widely with the highest turnover frequency observed on Hf-Beta (4.6 min−1), followed by Zr- and Sn-Beta (1.8 min−1 and 0.51 min−1, respectively). The lowest activation energy was observed on Hf-Beta (49.6 kJ mol−1) while Zr- and Sn-Beta showed higher but similar activation energy (60.7 and 60.4 kJ mol−1, respectively). The catalysts deactivate with time on stream (with a half-life of 3.5 hours or longer) and are poisoned by the reaction product furfuryl alcohol. Deactivation is caused partly by the polymerization of reactants and products that are retained in the zeolite pores and block access to active sites, but operation at higher temperature (408 K) slows the rate of deactivation significantly. The catalytic properties of the materials can be regenerated by calcination in air.

 Please wait while we load your content...
Please wait while we load your content...