Antagonistic effects leading to turn-on electrochemiluminescence in thermoresponsive hydrogel films†
Abstract
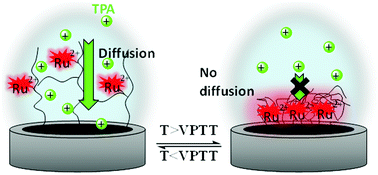
We report the effects of the swell-to-collapse transition on the electrochemical and electrochemiluminescence (ECL) properties of thermoresponsive pNIPAM films incorporating covalently-attached Ru(bpy)32+ luminophores. Upon the collapse of the film, the number of electrochemically-active Ru(bpy)32+ centers increases, due to the reduced distance between adjacent redox centers. To generate ECL, cationic and anionic coreactants are employed, which are free to diffuse in the medium. In both cases, the ECL intensity exhibits a remarkable amplification of up to 58-fold at the swell-to-collapse transition, whereas the oxidation current undergoes a steep decrease in the case of the cationic coreactant. Indeed, the diffusion of the coreactant is hindered by the collapse of the film. The fact that ECL emission is enhanced whereas less coreactant radicals are generated is an intriguing observation because it is classically believed that the higher the oxidation rate of the coreactant the stronger the ECL emission. Thanks to the study of such films where antagonistic effects occur, we demonstrate that ECL enhancement is neither correlated with the oxidation efficiency of the coreactant nor with the hydrophilic–hydrophobic transition, but solely due to film shrinking. The decrease of the distance between adjacent redox centers, which favors better electron-transfer processes, is found to be the main parameter governing the ECL enhancement. Finally, it is noteworthy that the present turn-on ECL signal with increasing temperature is unique compared to many thermoresponsive luminescent systems.

 Please wait while we load your content...
Please wait while we load your content...