Charge compensation assisted enhancement of photoluminescence in combustion derived Li+ co-doped cubic ZrO2:Eu3+ nanophosphors
Abstract
Red light emitting cubic Zr0.99Eu0.01O2:Li+ (0–9 mol%) nanoparticles are synthesized by a low temperature, self-propagating solution combustion method using oxalyl di-hydrazide (ODH) as fuel. In this study, we report systematic investigation of the effect of lithium ion (Li+) concentration on the structural properties and the photoluminescence of zirconia. With increasing lithium concentration, the crystallinity of the samples increases and the lattice strain decreases. The higher crystallinity is likely due to charge compensation achieved by replacing one Zr4+ ion by a Eu3+ and a Li+ ion. Scanning electron micrographs (SEM) reveal a mesoporous structure characteristic of combustion derived nanomaterials. Photoluminescence (PL) spectra show that the intensity of the red emission (606 nm) is highly dependent on Li+ ion concentration. Furthermore there is a promising enhancement in the associated lifetime. Upon Li+ doping, the PL intensity of the samples is found to increase by two fold compared to the undoped sample. Variation of PL intensity with Li+ concentration is attributed to the differences in probability of non-radiative recombination (relaxing). Intensity parameters (Ω2, Ω) and radiative properties such as transition rates (A), branching ratios (β), stimulated emission cross-section (σe), gain bandwidth (σe × Δλeff) and optical gain (σe × τ) are calculated using the Judd–Ofelt theory. The calculated values suggest that in optimally co-doped samples, in addition to improved crystallinity and charge compensation, the lowering of Eu3+ site symmetry and the increase in the covalency of Eu–O bonding due to interstitial Li are responsible for the observed enhancement in PL intensity.
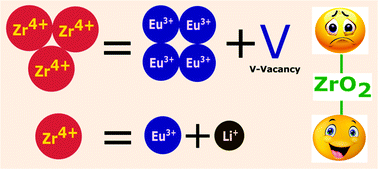


 Please wait while we load your content...
Please wait while we load your content...