Molecular dynamics study of di-CF4 based reverse micelles in supercritical CO2†
Abstract
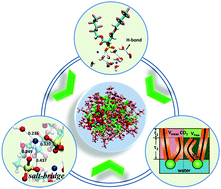
Reverse micelles (RMs) in supercritical CO2 (scCO2) are promising alternatives for organic solvents, especially when both polar and non-polar components are involved. Fluorinated surfactants, particularly double-chain fluorocarbon surfactants, are able to form well-structured RMs in scCO2. The inherent self-assembly mechanisms of surfactants in scCO2 are still subject to discussion. In this study, molecular dynamics simulations are performed to investigate the self-aggregation behavior of di-CF4 based RMs in scCO2, and stable and spherical RMs are formed. The dynamics process and the self-assembly structure in the RMs reveal a three-step mechanism to form the RMs, that is, small RMs, rod-like RMs and fusion of the rod-like RMs. Hydrogen-bonds between headgroups and water molecules, and salt bridges linking Na+ ions, headgroups and water molecules enhance the interfacial packing efficiency of the surfactant. The results show that di-CF4 molecules have a high surfactant coverage at the RM interface, implying a high CO2-philicity. This mainly results from bending of the short chain (C–COO–CH2–(CF2)3–CF3) due to the flexible carboxyl group. The microscopic insight provided in this study is helpful in understanding surfactant self-assembly phenomena and designing new CO2-philic surfactants.

 Please wait while we load your content...
Please wait while we load your content...