Combined TGA-MS kinetic analysis of multistep processes. Thermal decomposition and ceramification of polysilazane and polysiloxane preceramic polymers†
Abstract
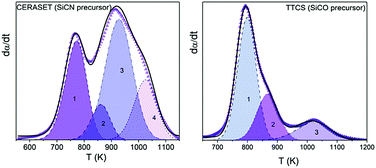
The polymer-to-ceramic transformation kinetics of two widely employed ceramic precursors, 1,3,5,7-tetramethyl-1,3,5,7-tetravinylcyclotetrasiloxane (TTCS) and polyureamethylvinylsilazane (CERASET), have been investigated using coupled thermogravimetry and mass spectrometry (TG-MS), Raman, XRD and FTIR. The thermally induced decomposition of the pre-ceramic polymer is the critical step in the synthesis of polymer derived ceramics (PDCs) and accurate kinetic modeling is key to attaining a complete understanding of the underlying process and to attempt any behavior predictions. However, obtaining a precise kinetic description of processes of such complexity, consisting of several largely overlapping physico-chemical processes comprising the cleavage of the starting polymeric network and the release of organic moieties, is extremely difficult. Here, by using the evolved gases detected by MS as a guide it has been possible to determine the number of steps that compose the overall process, which was subsequently resolved using a semiempirical deconvolution method based on the Frasier–Suzuki function. Such a function is more appropriate that the more usual Gaussian or Lorentzian functions since it takes into account the intrinsic asymmetry of kinetic curves. Then, the kinetic parameters of each constituent step were independently determined using both model-free and model-fitting procedures, and it was found that the processes obey mostly diffusion models which can be attributed to the diffusion of the released gases through the solid matrix. The validity of the obtained kinetic parameters was tested not only by the successful reconstruction of the original experimental curves, but also by predicting the kinetic curves of the overall processes yielded by different thermal schedules and by a mixed TTCS-CERASET precursor.

 Please wait while we load your content...
Please wait while we load your content...