Understanding the molecular switching properties of octaphyrins†
Abstract
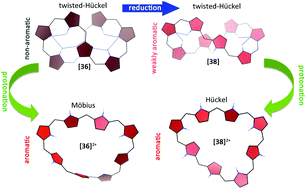
Several expanded porphyrins switch between Hückel, Möbius and twisted-Hückel topologies, encoding different aromaticity and NLO properties. Since the topological switch can be induced by different external stimuli, expanded porphyrins represent a promising platform to develop molecular switches for molecular electronic devices. In order to determine the optimum conditions for efficient molecular switches from octaphyrins, we have carried out a comprehensive quantum chemical study focusing on the conformational preferences and aromaticity of [36]octaphyrins. Different external stimuli for triggering the topological switch have been considered in our work, such as protonation and redox reactions. Importantly, the structure–property relationships between the molecular conformation, the number of π-electrons and aromaticity in octaphyrins have been established by using energetic, magnetic, structural and reactivity descriptors. Remarkably, we found that the aromaticity of octaphyrins is highly dependent on the π-conjugation topology and the number of π-electrons and it can be modulated by protonation and redox reactions. A non-aromatic figure-eight conformation is strongly preferred by neutral [36]octaphyrins that switches to a Möbius aromatic conformation upon protonation. Such a change of topology involves an aromaticity switch in a single molecule and is accompanied by a drastic change in the NLO properties. In contrast, the twisted-Hückel topology remains the most stable one in the oxidized and reduced species, but the aromaticity is totally reversed upon redox reactions. Aromaticity is shown to be a key concept in expanded porphyrins, determining the electronic, magnetic and NLO properties of these macrocycles.
- This article is part of the themed collection: Electron delocalization and aromaticity: 150 years of the Kekulé benzene structure

 Please wait while we load your content...
Please wait while we load your content...