Ultrafine Ag/polyoxometalate-doped AgCl nanoparticles in metal–organic framework as efficient photocatalysts under visible light†
Abstract
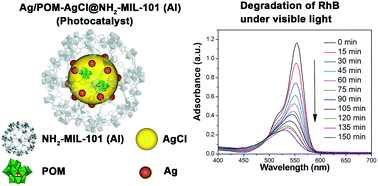
A new composite material of ultrafine Ag/PW11O39-doped AgCl nanoparticles (NPs) encapsulated in a metal–organic framework (NH2-MIL-101(Al)) was prepared by an incipient wetness impregnation method together with photo-reduction under UV irradiation. The as-prepared composite material (abbr. Ag/POM-AgCl@NH2-MIL-101(Al)) was characterized by XRD, EDX, SEM, XPS, FT-IR and diffuse reflectance spectroscopy (DRS). By virtue of the coordination interaction between the polyoxometalate (POM) unit and Ag+ ion in the K6[α-AgPW11O39] precursor and the confined cavity of NH2-MIL-101(Al), the fast reaction between Ag+ and Cl− ions was depressed and the over agglomeration of crystalline AgCl was limited. When the partial Ag+ ions were reduced into Ag0 metal in the AgCl NPs under UV irradiation, the resultant ultrafine Ag/POM-AgCl nanoparticles (NPs) possess an average particle size of 3.2 nm in diameter, representing one of the currently smallest Ag/AgCl composite NPs. The catalytic activity of this material was evaluated by the catalytic reduction of 4-nitrophenol and photocatalytic degradation of Rhodamine B (RhB) under visible light irradiation. Compared with its counterparts (Ag/AgCl NPs@NH2-MIL-101(Al)), the introduction of POM units into the Ag/AgCl composite NPs significantly improved the photocatalytic activity for degradation of RhB under visible light irradiation. A relevant photocatalytic mechanism was proposed.

 Please wait while we load your content...
Please wait while we load your content...