Fabrication of different copper nanostructures on indium-tin-oxide electrodes: shape dependent electrocatalytic activity†
Abstract
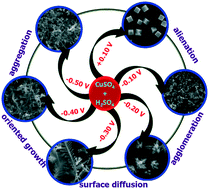
This paper describes the fabrication of cubic, spherical, dendritic and prickly copper nanostructures (CuNS) on indium-tin-oxide (ITO) substrates by electrodeposition and their electrocatalytic activity towards the oxidation of glucose and hydrazine. CuNS with different shapes were fabricated on ITO substrates by using different applied potentials of +0.10, −0.10, −0.30 and −0.50 V for 400 s in the presence of 10 mM CuSO4 and 0.1 M H2SO4. The formed CuNS were characterized by scanning electron microscopy (SEM), X-ray diffraction (XRD), energy dispersive X-ray analysis (EDAX), X-ray photoelectron spectroscopy (XPS) and electrical impedance spectroscopy (EIS). SEM images showed that cubic, spherical, dendritic and prickly CuNS were formed at applied potentials of +0.10, −0.10, −0.30 and −0.50 V, respectively. XPS showed characteristic peaks at 935 and 955 eV corresponding to Cu(0). The dendritic CuNS modified electrode exhibits a higher heterogeneous electron transfer rate constant of 3.70 × 10−7 cm s−1 and an electroactive area of 51.32 cm2 when compared to other CuNS modified electrodes. Further, the electrocatalytic activity of the different shaped CuNS modified ITO electrodes was examined towards the oxidation of glucose and hydrazine. Interestingly, the dendritic CuNS modified ITO substrate dramatically enhanced the oxidation currents of both glucose and hydrazine and also shifted their oxidation potential towards less positive potential when compared to bare ITO and other CuNS modified ITO substrates. The formation of dendritic CuNS was optimized with respect to deposition time, anion and pH and was monitored by SEM. Based on the results, a plausible mechanism for dendritic CuNS formation was proposed.

 Please wait while we load your content...
Please wait while we load your content...