Structural diversity of a series of coordination polymers built from 5-substituted isophthalic acid with or without a methyl-functionalized N-donor ligand†
Abstract
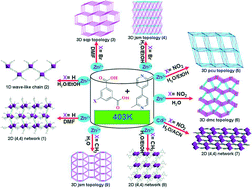
The reaction of X-H2BDC (X = H, Br, NO2 and CH3) and methyl-functionalized dmbpy ligands with metal salts under different solvent media conditions generates nine structurally diverse zinc(II)/cadmium(II)-based coordination polymers (CPs), {[Zn2(BDC)2(DMF)3]·DMF}n (1), [Zn(BDC)2(dmbpy)]n (2), {[(Me2NH2)2Zn3(Br-BDC)4]·3DMF·3H2O}n (3), [Zn2(Br-BDC)2(dmbpy)]n (4), {[Zn13(NO2-BDC)8(μ3-OH)2(μ2-OH)6(H2O)4]·12H2O·2NO3}n (5), [Zn2(NO2-BDC)2(dmbpy)(H2O)2]n (6), {[Cd(NO2-BDC)(dmbpy)0.5(H2O)]·(ACN)·H2O}n (7), [Zn(CH3-BDC)(dmbpy)0.5(H2O)]n (8), and [Zn(CH3-BDC)(dmbpy)0.5]n (9) (H2BDC = isophthalic acid, Br-H2BDC = 5-bromoisophthalic acid, NO2-H2BDC = 5-nitroisophthalic acid, CH3-H2BDC = 5-methylisophthalic acid, dmbpy = 2,2′-dimethyl-4,4′-bipyridine, DMF = N,N-dimethylformamide, ACN = acetonitrile). All the compounds are synthesized using dual linkers (X-H2BDC and dmbpy). 1 and 7–8 show a 2D (4,4) network when dinuclear metal ions and ligands are regarded as nodes and linkers, respectively. 2 is a wave-like chain with BDC ligands alternately pointing up and down. CPs 3–6 and 9 all exhibit 3D networks with 5-connected sqp, 6-connected jsm, 6-connected pcu, (3,4)-connected dmc and 6-connected jsm topologies, respectively. Furthermore, luminescence, thermogravimetric and chemical stability properties of these CPs were investigated. The results suggest that both organic ligands and solvent media influence the final resulting CPs.

 Please wait while we load your content...
Please wait while we load your content...