Identification of mesenchymal stem cell differentiation state using dual-micropore microfluidic impedance flow cytometry†
Abstract
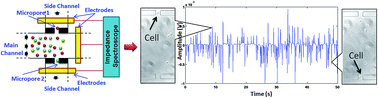
As stem cell therapies become more common in the clinic, there is a greater need for real-time, label-free monitoring of the differentiation status of the cells. In this paper, we present a dual-micropore-based, high-throughput microfluidic electrical impedance flow cytometer for non-invasive identification of the differentiation state of mesenchymal stem cells. The mesenchymal stem cells were induced to differentiate into osteoblasts over a 21 day period. Samples of mesenchymal stem cells and osteoblasts were flowed through the device, and impedance measurements were acquired over a frequency range from 50 kHz to 10 MHz. The opacity and relative angle, which shed light on the membrane capacitance and interior dielectric properties of cells, were used as interrogation parameters to analyze collected impedance data. Specifically, identification of mesenchymal stem cells and osteoblasts in a mixed population was optimized using a combination of opacity signature at 500 kHz and relative angle at 3 MHz. Identification of both cell populations in a mixed sample was successfully achieved with an accuracy of 87%. The results show a progressive increase in the number of osteoblasts throughout the 21 day differentiation process, with 36% more mesenchymal stem cells differentiated after 14 days of induction compared to after just 7 days. The dual-micropore microfluidic impedance flow cytometer system may become an important non-invasive tool for assessing stem cell quality and differentiation stages for future regenerative medicine applications.

 Please wait while we load your content...
Please wait while we load your content...