Detection of evoked acetylcholine release in mouse brain slices
Abstract
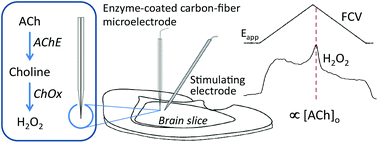
The study of transmitter interactions in reward and motor pathways in the brain, including the striatum, requires methodology to detect stimulus-driven neurotransmitter release events. Such methods exist for dopamine, and have contributed to the understanding of local and behavioral factors that regulate dopamine release. However, factors that regulate release of another key transmitter in these pathways, acetylcholine (ACh), are unresolved, in part because of limited temporal and spatial resolution of current detection methods. We have optimized a voltammetric method for detection of local stimulus-evoked ACh release using enzyme-coated carbon-fiber microelectrodes and fast-scan cyclic voltammetry. These electrodes are based on the detection of H2O2 generated by the actions of acetylcholine esterase and choline oxidase, and reliably respond to ACh in a concentration-dependent manner. Methods for enzyme coating were optimized for mechanical stability that allowed for their use in ex vivo brain slices. We report here the first quantitative assessment of extracellular ACh concentration after local electrical stimulation in dorsal striatum in slices from control mice. The selective detection of ACh under these conditions was confirmed by showing that the response detected in the control slices was absent in slices from mice bred to lack ACh synthesis in the forebrain. These electrodes represent a new tool to study ACh and ACh–dopamine interactions with micrometer spatial resolution.
- This article is part of the themed collection: In memory of Craig Lunte


 Please wait while we load your content...
Please wait while we load your content...