A one-step colorimetric acid–base titration sensor using a complementary color changing coordination system†
Abstract
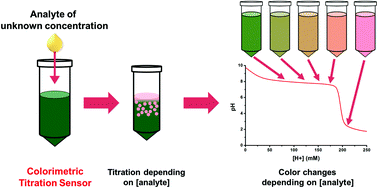
We report the development of a colorimetric sensor that allows for the quantitative measurement of the acid content via acid–base titration in a single-step. In order to create the sensor, we used a cobalt coordination system (Co-complex sensor) that changes from greenish blue colored Co(H2O)4(OH)2 to pink colored Co(H2O)62+ after neutralization. Greenish blue and pink are two complementary colors with a strong contrast. As a certain amount of acid is introduced to the Co-complex sensor, a portion of greenish blue colored Co(H2O)4(OH)2 changes to pink colored Co(H2O)62+, producing a different color. As the ratio of greenish blue and pink in the Co-complex sensor is determined by the amount of neutralization reaction occurring between Co(H2O)4(OH)2 and an acid, the sensor produced a spectrum of green, yellow green, brown, orange, and pink colors depending on the acid content. In contrast, the color change appeared only beyond the end point for normal acid–base titration. When we mixed this Co-complex sensor with different concentrations of citric acid, tartaric acid, and malic acid, three representative organic acids in fruits, we observed distinct color changes for each sample. This color change could also be observed in real fruit juice. When we treated the Co-complex sensor with real tangerine juice, it generated diverse colors depending on the concentration of citric acid in each sample. These results provide a new angle on simple but quantitative measurements of analytes for on-site usage in various applications, such as in food, farms, and the drug industry.
- This article is part of the themed collection: Emerging Investigators

 Please wait while we load your content...
Please wait while we load your content...