Synthesis and analysis of novel analogues of dexrazoxane and its open-ring hydrolysis product for protection against anthracycline cardiotoxicity in vitro and in vivo
Abstract
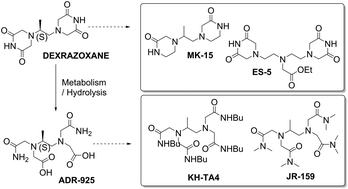
Cardiotoxicity is a serious drawback of anthracycline anti-cancer drugs and dexrazoxane is the only cardioprotective agent with clinically established efficacy. Iron-mediated oxidative stress is traditionally believed to be the primary cause of anthracycline cardiotoxicity, and dexrazoxane-induced cardioprotection is attributed to iron chelating properties of its open ring metabolite, ADR-925, which may inhibit the oxidative injury. However, dexrazoxane is also a catalytic inhibitor of topoisomerase II (TOP2), and the role of oxidative stress in clinically relevant forms of cardiotoxicity has increasingly been questioned. In this study, novel analogues of dexrazoxane (MK-15, ES-5) and ADR-925 (KH-TA4, JR-159) were synthesized, and evaluated in vitro and in vivo. When examined in the leukemic cell line, HL-60, these novel analogues did not interfere with the anti-proliferative action of daunorubicin. In contrast to dexrazoxane, they had no anti-cancer effect on their own and the changes in the chemical structure resulted in a loss of TOP2 inhibitory activity. Although some of the novel compounds showed significant anti-oxidant and iron chelating properties in vitro, they did not protect isolated cardiomyocytes and rabbits from daunorubicin-induced cardiotoxicity and heart failure. Importantly, dexrazoxane has been found to be a relatively weak intracellular iron chelator and it failed to protect the isolated cardiomyocytes from model oxidative injury induced by hydrogen peroxide. However, in contrast to all novel analogues, dexrazoxane induced depletion of the TOP2 beta isoform. This isoform is typical for terminally differentiated cells and its genetic deletion has been reported to overcome anthracycline-induced cardiotoxicity. Hence, TOP2 beta, rather than (or along with) iron chelation, may be a promising target for effective cardioprotection induced by bisdioxopiperazine agents.

 Please wait while we load your content...
Please wait while we load your content...