Optical and electronic properties of air-stable organoboron compounds with strongly electron-accepting bis(fluoromesityl)boryl groups†
Abstract
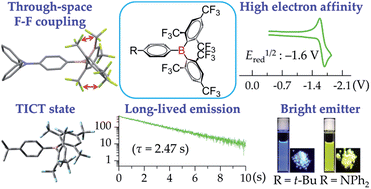
Three compounds with phenyl (1), 4-tert-butylphenyl (2) and 4-N,N-diphenylaminophenyl (3) groups attached to bis(fluoromesityl)boryl ((FMes)2B) through B–C bonds have been prepared. The restricted rotation about the B–C bonds of boron-bonded aryl rings in solution has been studied by variable-temperature 19F NMR spectroscopy, and through-space F–F coupling has been observed for 3 at low temperature. Steric congestion inhibits binding of 1 by Lewis bases DABCO and tBu3P and the activation of H2 in their presence. Photophysical and electrochemical studies have been carried out on 2, 3, and an analogue of 3 containing a bis(mesityl)boryl ((Mes)2B) group, namely 4. Both 2 and 3 show bright emission in nonpolar solvents and in the solid-state, very strong electron-accepting ability as measured by cyclic voltammetry, and good air-stability. In addition, 2 displayed unusually long-lived emission (τ = 2.47 s) in 2-MeTHF at 77 K. The much stronger acceptor strength of (FMes)2B than (Mes)2B leads to significantly red-shifted emission in solution and the solid state, stronger emission solvatochromism, and significantly lower reduction potentials. Theoretical calculations confirm that 2 and 3 tend to form highly twisted excited states with good conjugation between one FMes group and the boron atom, which correlate well with their blue-shifted solid-state emissions and low kr values in solution.

 Please wait while we load your content...
Please wait while we load your content...