Highly emissive excited-state intramolecular proton transfer (ESIPT) inspired 2-(2′-hydroxy)benzothiazole–fluorene motifs: spectroscopic and photophysical properties investigation†
Abstract
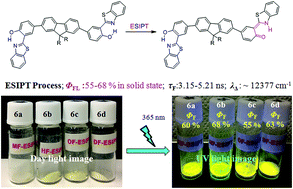
Tuning or switching of the solid state luminescence of organic materials is an attractive target for both basic research and practical applications. In the present study, solid state emissive compounds with very high quantum efficiencies (ΦF up to 68%) were achieved by chemical alteration of the excited state intramolecular proton transfer (ESIPT) 2-2′-hydroxy benzothiazole (HBT) unit. Five ESIPT inspired compounds based on fluorene were synthesized via Suzuki coupling reaction. Their photophysical properties were studied by means of steady state absorption, emission spectra and a time resolved emission method in solid as well as in solution of different polarities. The fluorophores showed absorption in the UV region and emission in the visible region with large Stokes shift (∼232 nm). Efficient yellow emissive compounds showed very high quantum yields (ΦF = 55–68%) in the solid state, which are the highest quantum yields in the solid state to the best of our knowledge, for fluorene based ESIPT molecules. The fluorescence lifetime in the solid state is between 3.48–5.21 ns, while it is 5–10 fold less in chloroform (0.52–0.75 ns) solution. The optical properties of these compounds are sensitive towards the polarity of the medium. The structural properties, such as X-ray single crystal analyses, DSC and TGA were studied, and the lack of stacking and/or hydrogen bonding interactions around HBT motifs reveals enough room for ESIPT in the series of molecules even in their solid state. The DFT computations were performed to support experimental results and the calculations are well in line with the experimental results. These suggest high quantum efficiency ascribed to the large orbital energy difference between HOMOs and LUMOs of enol and keto forms transformed via ESIPT, and hence, singlet energy localization onto the keto form. The intra-molecular charge transfer nature between fluorene and HBT units plays a key role for the localization of energy on HBT motifs in their excited states.


 Please wait while we load your content...
Please wait while we load your content...