Sustainable, eco-friendly polyesters synthesized from renewable resources: preparation and thermal characteristics of poly(dimethyl-propylene furanoate)
Abstract
Poly(2,2-dimethyl-1,3-propylene furanoate) (PDMPF), an interesting sustainable biobased polyester based on 2,5-furan dicarboxylic acid (FDCA), was synthesized by applying the two-stage melt polycondensation method. The polyester exhibited a melting temperature of Tm = 198 °C and a glass transition temperature of Tg = 68 °C. Multiple melting was observed for the samples crystallized isothermally at temperatures ranging from 160 to 175 °C. Extensive recrystallization was evidenced by modulated temperature differential scanning calorimetry (MDSC) during heating. The equilibrium melting temperature was found to be 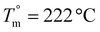 and the enthalpy of fusion of the pure crystalline polymer was ΔHf = 133 J g−1. The crystallization rates were analyzed according to the secondary nucleation theory, and a relatively large nucleation constant Kg was obtained, representing the rigidity of the macromolecular chains. Large spherulites were observed during isothermal crystallization tests with the aid of polarized light optical microscopy (PLOM). The polyester showed significant stability during the thermal degradation tests. Finally, the degradation mechanism was investigated by employing a pyrolyzer–gas chromatography–mass spectroscopy (Py-GC-MS) system.
and the enthalpy of fusion of the pure crystalline polymer was ΔHf = 133 J g−1. The crystallization rates were analyzed according to the secondary nucleation theory, and a relatively large nucleation constant Kg was obtained, representing the rigidity of the macromolecular chains. Large spherulites were observed during isothermal crystallization tests with the aid of polarized light optical microscopy (PLOM). The polyester showed significant stability during the thermal degradation tests. Finally, the degradation mechanism was investigated by employing a pyrolyzer–gas chromatography–mass spectroscopy (Py-GC-MS) system.


 Please wait while we load your content...
Please wait while we load your content...