Dual side chain control in the synthesis of novel sequence-defined oligomers through the Ugi four-component reaction†
Abstract
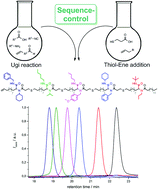
The synthesis of sequence-defined oligomers by the iterative application of the modular Ugi four-component reaction (U-4CR) and the efficient thiol–ene addition reaction is described. By varying the amine component in the U-4CR, a sequence-defined and monodisperse tetramer (M = 1568.5 g mol−1) was obtained. More interestingly, if both the amine and the isocyanide components were varied simultaneously in the U-4CR, a double sequence-controlled, monodisperse pentamer (M = 2411.8 g mol−1) bearing ten different and selectable side chains was obtained. All oligomers were thoroughly characterized by NMR and IR spectroscopy, mass spectrometry and GPC.

 Please wait while we load your content...
Please wait while we load your content...