Copper-catalysed cross-coupling: an untapped potential
Abstract
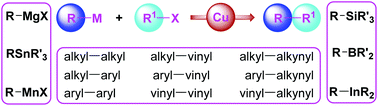
Copper is emerging as a viable catalytic metal for cross-coupling reactions to construct carbon–carbon (C–C) bonds. Recent revelations that Cu-catalysts can execute with high efficacy the cross-couplings of a variety of organometallic reagents, including organomagnesium, organoboron, organosilicon, organoindium and organomanganese, with alkyl, aryl and heteroaryl halides clearly demonstrate the versatility of Cu-based catalytic systems in conducting these reactions. In addition, Cu-catalysts are exhibiting a unique reactivity pattern that allows ligandless cross-coupling for aryl–heteroaryl and heteroaryl–heteroaryl bond formation, a transformation that generally requires special custom-designed ligands with Pd-catalysts. This review summarises early discoveries and subsequent advancements made in the area of Cu-catalysed cross-couplings of organometallic reagents with organohalides to form C–C bonds.

 Please wait while we load your content...
Please wait while we load your content...