Tunable luminescence and energy transfer properties of a novel Na4Ca4Si6O18:Ce3+,Mn2+ phosphor†
Abstract
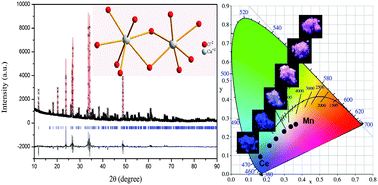
A series of color-tunable Na4Ca4Si6O18:Ce3+,Mn2+ phosphors were synthesized via a high-temperature solid-state reaction. The effect of Ce3+ concentration on the emission intensity of Na4Ca4Si6O18:Ce3+ is studied, and the energy transfer from Ce3+ to Mn2+ is observed. The emission spectra of the Na4Ca4Si6O18:Ce3+,Mn2+ phosphors show a blue band at 430 nm of Ce3+ and a red band at 590 nm of Mn2+, originating from the allowed 5d → 4f transition of the Ce3+ ion and the 4T1g(4G) → 6A1g(6S) transition of the Mn2+ ion, respectively. Non-radiative transitions between the Ce3+ and Mn2+ ions in the Na4Ca4Si6O18 host are demonstrated to be dipole–quadrupole interactions. Results indicate that the varied emitted color from blue to red can be achieved by tuning the relative ratio of the Ce3+ to Mn2+ ions based on the principle of energy transfer. We have demonstrated that Na4Ca4Si6O18:Ce3+,Mn2+ phosphors can be promising candidates for color-tunable phosphors applied in near-UV white LEDs.

 Please wait while we load your content...
Please wait while we load your content...