Aberrant zinc binding to immature conformers of metal-free copper–zinc superoxide dismutase triggers amorphous aggregation†
Abstract
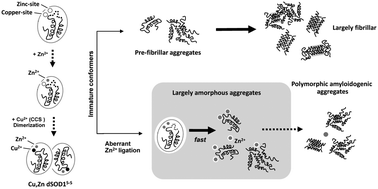
Superoxide dismutase 1 (SOD1) is a Cu/Zn metalloenzyme that aggregates in amyotrophic lateral sclerosis (ALS), a fatal neurodegenerative disorder. Correct metal insertion during SOD1 biosynthesis is critical to prevent misfolding; however Zn2+ can bind to the copper-site leading to an aberrantly metallated protein. These effects of Zn2+ misligation on SOD1 aggregation remain to be explored, even though Zn2+ levels are upregulated in ALS motor neurons. Here we use complementary biophysical methods to investigate Zn2+ binding and its effects on the aggregation of three immature metal-free SOD1 conformers that represent biogenesis intermediates: dimeric, monomeric and reduced monomeric SOD1. Using isothermal titration calorimetry we determined that Zn2+ binds to all conformers both at the zinc- as well as to the copper-site; however Zn2+ binding mechanisms to the zinc-site have distinct characteristics across immature conformers. We show that this ‘zinc overload’ of immature SOD1 promotes intermolecular interactions, as evidenced by dynamic light scattering and ThT fluorescence kinetic studies. Analysis of aged zinc-induced aggregates by energy-dispersive X-ray and electron energy-loss spectroscopy shows that aggregates integrate some Zn2+. In addition, electron diffraction analysis identifies nano-scaled crystalline materials and amyloid fibril-like reflections. Transmission electron microscopy reveals that Zn2+ diverts the SOD1 aggregation pathway from fibrils to amorphous aggregate, and electrophoretic analysis evidences an increase in insoluble materials. Overall, we provide evidence that aberrant zinc coordination to immature conformers broadens the population of SOD1 misfolded species at early aggregation stages and provide evidence for a high structural polymorphism and heterogeneity of SOD1 aggregates.
- This article is part of the themed collection: Zinc in the Biosciences


 Please wait while we load your content...
Please wait while we load your content...