Eco-friendly electrocatalytic oxidation of alcohols on a novel electro generated TEMPO-functionalized MCM-41 modified electrode†
Abstract
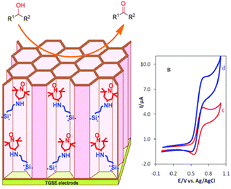
The development of a highly efficient and waste-free system for the selective oxidation of alcohols without using expensive transition metal catalysts is an extremely important and highly challenging area in organic chemistry and particularly in the chemical industry. In this study, a novel procedure for an environmentally friendly electrochemical oxidation of alcohols is introduced, in which a thin layer of ordered mesoporous silica (MCM-41) with well-oriented channels is constructed on an electrode surface and then functionalized with 2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO) as an electroactive organocatalyst. The electrocatalytic system operates efficiently in mild bicarbonate solution, at room temperature without the need for any co-catalyst, and provides a very fast, simple, selective and waste-free protocol for the oxidation of a wide variety of alcohols. The proposed modified electrodes could be easily employed on a 20 mmol scale in one reaction run and show very good stability and can be successfully reused for several runs. In terms of electrocatalytic activity, this system enables unprecedented turnover frequencies (TOF) of up to 3070 h−1, which is much superior to all the reported nitroxyl radicals under chemical, electrochemical, or aerobic oxidation conditions.


 Please wait while we load your content...
Please wait while we load your content...