Planar pentacoordinate carbons in CBe54− derivatives†
Abstract
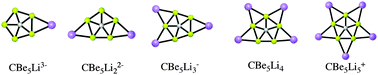
The potential energy surfaces of a series of clusters with formula CBe5Linn−4 (n = 1 to 5) have been systematically explored. Our computations show that the lithium cations preserve the CBe54− pentagon, such that the global minimum structure for these series of clusters has a planar pentacoordinate carbon (ppC) atom. The systems are primarily connected via a network of multicenter σ-bonds, in which the C atom acts as σ-acceptor and this acceptance of charge is balanced by the donation of the 2pz electrons to the π-cloud. The induced magnetic field analysis suggests that the clusters with formula CBe5Linn−4 (n = 1 to 5) are fully delocalized. The fact that these ppC-containing clusters are the lowest-energy forms on the corresponding potential energy surfaces raises expectations that these species can be prepared experimentally in the gas phase.

 Please wait while we load your content...
Please wait while we load your content...