Aspects of crystal engineering arising from packing behavior of functional mono para-substituted trityl compounds†
Abstract
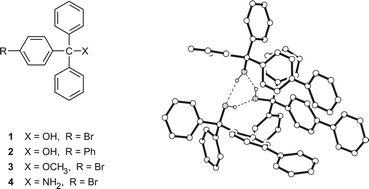
Four trityl compounds differing both in the functional group (OH, OMe, NH2) at the specific trityl carbon and a para substituent, being bromine or phenyl at one of the phenyl groups, have been prepared and structurally studied by means of single crystal X-ray diffraction and making use of Hirshfeld surface analysis. Compared to the structures of corresponding prototypes and analogues, specific interaction modes and packing motifs including cluster aggregates as well as non-cluster type structures depending on the substitution and involving polymorphism were found, opening potential trend prediction with reference to crystal engineering being useful in this compound class.

 Please wait while we load your content...
Please wait while we load your content...