Rh(iii)-catalyzed 7-azaindole synthesis via C–H activation/annulative coupling of aminopyridines with alkynes†
Abstract
An efficient Rh(III)-catalyzed 7-azaindole synthesis was developed via C–H activation/annulative coupling of aminopyridines with alkynes. The reaction was highly regioselective and tolerated various functional groups, permitting the construction of various 7-azaindoles.
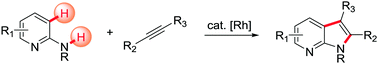

 Please wait while we load your content...
Please wait while we load your content...