A protocol for the quality assessment of illegally distributed human growth hormones with respect to identity, purity, endotoxin level and microorganism content
Abstract
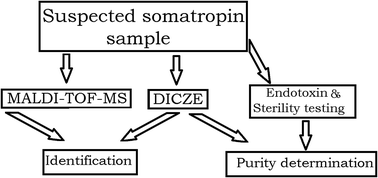
Different methods based on MALDI-TOF-MS and double injection capillary zone electrophoresis (DICZE) were used for the identification and purity determination of somatropin in illegally distributed products. During the past few years, more than 200 products suspected to contain somatropin have been analysed. Some of the samples were also subjected to control microorganisms and endotoxins. The identification of somatropin was carried out by peptide mapping using trypsin as the proteolytic enzyme. A double chain peptide cross-linked via a disulfide bond was used as the signature peptide. Capillary electrophoresis in double injection mode was applied to both identification and purity determination of the samples. The identification was based on the comparison between the observed migration time of the reference standard and the calculated migration time of the analyte, being present in the second injection plug. The DICZE provides electrophoretic fingerprints of intact somatropin and the related proteins which facilitate the identification. In addition, some of these samples revealed the presence of microorganisms as well as a high level of endotoxins. Taken together, the doubtful quality of the analysed samples and the microbiological findings represent a serious threat for the consumers and public health.

 Please wait while we load your content...
Please wait while we load your content...