Photoelectrochemical water splitting in an organic artificial leaf†
Abstract
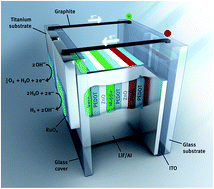
Photoelectrochemical water splitting is demonstrated in an organic artificial leaf composed of a triple junction polymer solar cell for light absorption and charge generation and low-overpotential catalytic electrodes for hydrogen and oxygen evolution. For small area solar cells (<0.1 cm2), a solar to hydrogen conversion efficiency of 5.4% is obtained using RuO2 catalysts. Using earth-abundant NiMoZn and Co3O4 catalysts for hydrogen and oxygen evolution, the efficiency is 4.9%. For larger area (1.7 cm2) solar cell devices the solar to hydrogen efficiency with RuO2 catalysts reduces to 3.6% as a consequence of an increased overpotential for water splitting. This shifts the operating point of the water splitting device beyond the maximum power point of the solar cell and reduces the photocurrent.

 Please wait while we load your content...
Please wait while we load your content...