SO42− anion directed hexagonal-prismatic cages via cooperative C–H⋯O hydrogen bonds†
Abstract
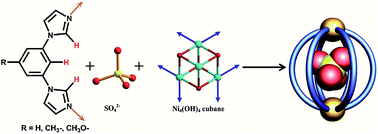
Hexagonal-prismatic cages are constructed from cubane-like Ni4(μ3-OH)4 clusters generated in situ and clip-like organic ligands. The SO42− anions act as the template for the formation of the above cages via weak cooperative C–H⋯O hydrogen bonds. Further research shows that three internal hydrogen atoms of the bib ligand or its derivatives are necessary because they are all involved in the cooperative C–H⋯O hydrogen bonding with the captured SO42− anions. More importantly, not only the size and shape but also the charge of the SO42− anions dominates the formation of the cages.

 Please wait while we load your content...
Please wait while we load your content...