Iron-catalyzed kinetic resolution of N-sulfonyl oxaziridines†
Abstract
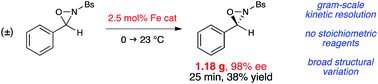
We have developed a highly selective kinetic resolution of N-sulfonyl oxaziridines. This reaction utilizes an inexpensive and easily synthesized iron bis(oxazoline) catalyst to promote the efficient rearrangement of oxaziridines to the corresponding N-sulfonyl imides; no sacrificial reagents are required to effect this resolution. This process is readily translated to gram scale, which provides a practical method for the preparation of structurally diverse, enantiopure N-sulfonyl oxaziridines for use as reagents in organic synthesis.

 Please wait while we load your content...
Please wait while we load your content...