Synthesis and characterization of fully biobased aromatic polyols – oxybutylation of condensed tannins towards new macromolecular architectures†
Abstract
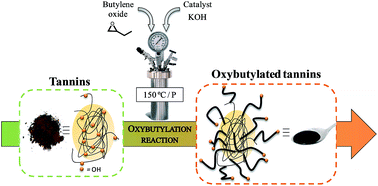
For the first time, different tannins were oxybutylated to obtain aromatic and fully biobased polyols. The main interest in using 1,2-butylene oxide (BO) instead of e.g., propylene oxide is because BO can be easily biobased and biosynthesized from biomass. BO can be obtained by epoxidation of butane, produced from biobased butanol, synthesized by biotechnological means. The oxybutylation was carried out using potassium hydroxide as a catalyst, in a high-pressure batch reactor, without solvent, in agreement with the main principles of green chemistry. Firstly, different biobased aromatic polyols were synthesized from gambier tannin (Uncaria gambir) and fully characterized. To better understand the conditions and mechanisms of the synthesis, the influence of the tannin/BO ratio was assessed through the evolution of the hydroxyl content, homopolymer content, molar mass distributions and the corresponding viscosities. In addition, the thermal properties were evaluated to better understand the final molecular architectures. Secondly, selected conditions of synthesis were applied to other types of tannins from pine (Pinus pinaster), mimosa (Acacia mearnsii) and quebracho (Schinopsis balansae and Schinopsis lorentzii). The corresponding results were compared to extend this study. For all tannins, the number and length of the grafted chains were carefully characterized. Great variations in reactivity have been found between the different botanical sources. Tailor-made, fully biobased and controlled aromatic macropolyols can be obtained by modulating the main parameters of the oxybutylation reaction and the type of botanical tannin source.

 Please wait while we load your content...
Please wait while we load your content...