A new aza-BODIPY based NIR region colorimetric and fluorescent chemodosimeter for fluoride†
Abstract
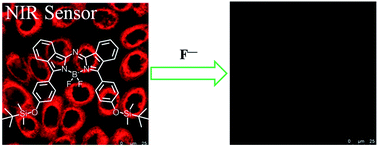
The synthesis and characterization of a novel NIR region fluoride sensor, that makes use of the aza-boron-dipyrromethene (aza-BODIPY) fluorophore, is described. An arylmagnesium bromide was formed by reacting 4-bromophenol with a tert-butyldimethylsilyl protecting group and magnesium, which was then used to prepare the aza-BODIPY through a reaction with phthalonitrile. The unusually strong affinity between the fluoride anion and silicon is used to create a sensor dye, which exhibits a highly specific, rapid colorimetric and ‘turn-off’ fluorescence response for F− in solution and in living HeLa cells. With F−, there is enhanced intramolecular charge transfer within the S1 state and this results in efficient nonradiative decay and hence in a marked decrease in fluorescence emission intensity.

 Please wait while we load your content...
Please wait while we load your content...