Terpolymerization of propylene oxide and vinyl oxides with CO2: copolymer cross-linking and surface modification via thiol–ene click chemistry
Abstract
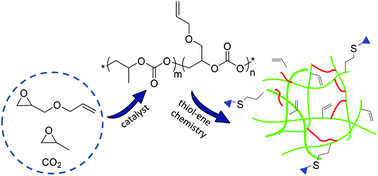
Terpolymerization reactions of epoxides bearing vinyl groups, propylene oxide and carbon dioxide catalyzed by binary and bifunctional (salen)Co(III) complexes have provided polycarbonates of various compositions depending on the feed ratios of the epoxide monomers. Fineman–Ross analysis of these polymerization reactions revealed reactivity ratios for propylene oxide (PO)/vinyl oxide (VIO)/CO2 of rPO = 3.74 and rVIO = 0.224 at 25 °C which reflect both the binding constants of the epoxides to the cobalt center as well as the rate constants for the ring-opening process. Less discrimination of epoxides was noted when carrying out the process using PO/allyl glycidyl ether (AGE)/CO2, where the reactivity parameters were rPO = 0.755 and rAGE = 0.876 at 25 °C. In order to enhance the polycarbonate's mechanical and thermal properties the terpolymers from PO/AGE/CO2, where the vinyl pendant groups are more randomly distributed in the polymeric material, were cross-linked via thiol–ene chemistry using two different thiols, ethylene glycol bis(3-mercaptoproionate) and pentaerythritol tetrakis(mercaptoacetate). FT-Raman spectroscopy provided evidence that successful cross-linking has occurred. Dynamic mechanical analysis (DMA) measurements on these terpolymers were performed as a function of the cross-link densities. Cross-linked films derived from the tetradentate cross-linker displayed a 50% increase in rubbery modulus between 50% and 100% C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond conversion; concomitantly, the Tg increased with increasing cross-link densities. In addition, the cross-linked films with 25% C
C bond conversion; concomitantly, the Tg increased with increasing cross-link densities. In addition, the cross-linked films with 25% C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond conversion were reacted with N-acetyl-L-cysteine and 2-(Boc-amino)ethanethiol, and confirmed by FTIR and XPS, to provide surface functionalized films containing carboxyl or amine groups for various applications.
C bond conversion were reacted with N-acetyl-L-cysteine and 2-(Boc-amino)ethanethiol, and confirmed by FTIR and XPS, to provide surface functionalized films containing carboxyl or amine groups for various applications.


 Please wait while we load your content...
Please wait while we load your content...