Synthesis and morphological studies of a poly(5,6-difluorobenzo-2,1,3-thiadiazole-4,7-diyl-alt-quaterchalcogenophene) copolymer with 7.3% polymer solar cell efficiency†
Abstract
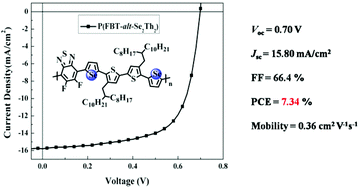
To obtain a poly(5,6-difluorobenzo-2,1,3-thiadiazole-4,7-diyl-alt-quaterchalcogenophene) (P(FBT-alt-CP4)) copolymer with a small optical band gap (Eg), and to achieve high short-circuit current (Jsc) in the P(FBT-alt-CP4) : PC71BM polymer solar cells (PSCs), P(FBT-alt-Se2Th2), which contains selenophene-2,5-diyl (–Se–) π-bridges, was synthesized. P(FBT-alt-Se2Th2) shows a Eg of 1.56 eV and is strongly aggregated in solution. Wide angle X-ray diffraction (WAXD) and grazing incidence X-ray diffraction (GI-XRD) results revealed the high solid-state order of P(FBT-alt-Se2Th2) and its edge-on orientation on the substrate. It delivered a high hole mobility (μh) of 0.36 cm2 V−1 s−1 in organic field-effect transistors (OFETs). The strong aggregation tendency of P(FBT-alt-Se2Th2) caused large segregation domains in the P(FBT-alt-Se2Th2) : PC71BM thin film, as is seen in the high-resolution transmission electron microscopy (HR-TEM) images. The addition of 8 vol% of 1-chloronaphthalene (1-CN) effectively suppressed the aggregation and led to more homogeneous active layer morphology. The improved morphology enhanced the Jsc of the PSCs. A superior PCE of 7.34% with a Voc of 0.70 V, a Jsc of 15.8 mA cm−2, and a FF of 66.4% was achieved in the inverted P(FBT-alt-Se2Th2) : PC71BM PSCs. The strong aggregation of P(FBT-alt-Se2Th2) is likely related to its more straight conjugated backbone according to the theoretical calculation results of the FBT-alt-Se2Th2 repeat unit.

 Please wait while we load your content...
Please wait while we load your content...