Highly stable hybrid selenophene-3,4-ethylenedioxythiophene as electrically conducting and electrochromic polymers†
Abstract
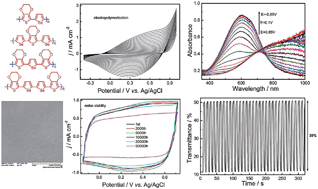
A family of four novel selenophene–EDOT oligomers were synthesized using Stille coupling and electropolymerized to form highly stable conducting hybrid polymers with excellent electrochromic properties. Structure–property relationships of the oligomers and hybrid polymers, including electrochemical, electronic and optical properties, quantum chemistry calculations and morphology, were systematically explored. The oligomer precursors with planar structures exhibit blue to orange emission characteristics with quantum yields ranging from 1.5 to 9.0%; they may be used as building blocks for the rational design of fluorescent conjugated systems with enhanced main chain planarity. Cyclic voltammetry shows low oxidation potentials ranging from 0.53 to 0.89 V vs. Ag/AgCl, leading to the facile electrodeposition of uniform hybrid polymer films with outstanding electroactivity and stability at low oxidation potentials. The obtained hybrid polymers featured the combined advantages of polyselenophene and PEDOT, such as the lower band gap and better planarity of polyselenophene and the high conductivity, transparency and excellent stability of PEDOT. The hybrid polymers show planar π-conjugated backbones with band gaps ranging from 1.54 to 1.75 eV. They are electrochromic in nature with colour changing from purplish, reddish and saturated blue in the reduced form to transparent sky blue/green upon oxidation. Further kinetic studies demonstrated that the hybrid polymers have decent contrast ratios (22–36%), favourable coloration efficiencies (∼200 cm2 C−1), low switching voltages, fast response time (0.5 s), excellent stability and colour persistence. These materials provide more plentiful electrochromic colours and hold promise for display applications.

 Please wait while we load your content...
Please wait while we load your content...