Transition-metal-free oxidative carboazidation of acrylamides via cascade C–N and C–C bond-forming reactions†
Abstract
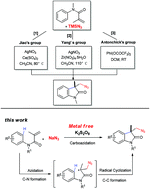
A novel transition-metal-free oxidative carboazidation of acrylamides using inexpensive NaN3 and K2S2O8 was achieved, which not only provided an efficient method to prepare various N3-substituted oxindoles, but also represented a novel strategy for C–N and C–C bond formation via a free-radical cascade process. This transformation exhibits excellent functional group tolerance, affording the desired oxindoles in good to excellent yields.

 Please wait while we load your content...
Please wait while we load your content...