Synthesis of macrolactam analogues of radicicol and their binding to heat shock protein Hsp90†
Abstract
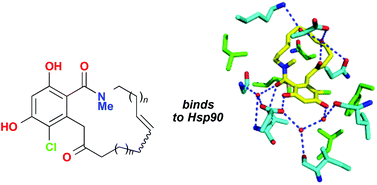
A series of macrolactam analogues of the naturally occurring resorcylic acid lactone radicicol have been synthesised from methyl orsellinate in 7 steps, involving chlorination, protection of the two phenolic groups, and hydrolysis to the benzoic acid. Formation of the dianion and quenching with a Weinreb amide results in acylation of the toluene methyl group that is followed by amide formation and ring closing metathesis to form the macrocyclic lactam. Final deprotection of the phenolic groups gives the desired macrolactams whose binding to the N-terminal domain of yeast Hsp90 was studied by isothermal titration calorimetry and protein X-ray crystallography.

 Please wait while we load your content...
Please wait while we load your content...