Studies of the complexation behavior of tetramorpholinylo-PNP-lariat ether with Ag(i), Ca(ii), Cd(ii), Cu(ii) and Pb(ii) using Electrospray Ionization Mass Spectrometry
Abstract
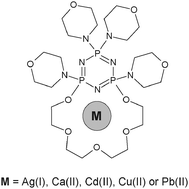
In this publication the cationic metal complexes of tetramorpholinylo-PNP-lariat ether have been studied using electrospray ionization mass spectrometry (ESI-MS). The tandem mass spectra (MS/MS) of these complexes have also been tested to evaluate the stability of the different types of the complexes formed. As occurred, all selected metal cations form the complexes with 1 : 1 stoichiometry with the investigated ligand. Only silver ions create a “sandwich” type complex. Furthermore, the divalent cations form complexes with the nitrate anion adduct. In the case of Ca(II), Cd(II) and Pb(II) we also observed another type of the species with an additional water molecule attached to the parent complex.

 Please wait while we load your content...
Please wait while we load your content...